Since 2008, I have been working on this other crucial step in gene expression that takes place downstream of mRNA 3’-end formation. The obvious initial connection between my present postdoctoral position and my Ph.D. work was that Nab2p is also involved in mRNA export from the nucleus to the cytoplasm in yeast 6.
Since 2000, it has been shown by various research groups that the proteins deposited on the mRNA during the maturation stage serve as adaptors for the heterodimeric mRNA export receptor Nxf1:p15 (also called TAP:p15, or Nxf1:Nxt1 ; Mex67p:Mtr2p in budding yeast): among them are canonical adaptors, splicing factors, and 3'-end processing factors 7-13. Their presence on the mRNA also stand for a hallmark of completion of the maturation step and therefore are part of a quality control deciding the fate of the mRNA: export to the cytoplasm for translation into proteins or nuclear retention and destruction by the nuclear exosome 14,15. Initial studies from various research groups suggested that the adaptor proteins were somehow bridging the RNA and the mRNA export receptor Nxf1 8,16. However, Dr Guillaume Hautbergue from the Wilson lab has actually shown that although the mRNA is initially bound to the adaptor, it is ultimately handed over to Nxf1. Upon binding of the adaptor to Nxf1, two concomitant phenomenon take place: the RNA is released from the adaptor by means of mutually exclusive interactions, and Nxf1's RNA binding activity is stimulated by an unknown mechanism 11. This allows efficient transfer of mRNAs to the mRNA export receptor Nxf1 that will finally contact the nucleopore and allow the mRNP to be translocated to the cytoplasm.
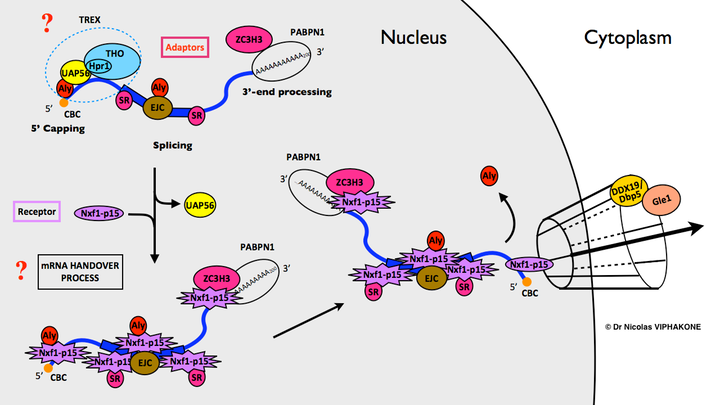
Summary of the steps licensing the messenger RNA for export from the nucleus to the cytoplasm, as known in 2008. Questions marks indicate that the precise molecular function of the THO-TREX complex and the molecular mechanism of the mRNA handover to the export receptor Nxf1:p15 were unknown at that time. For simplicity, the involvement of the TREX-2 complex and other regulations have been omitted. More up to date version in our review.
In this context, my current work in the Wilson lab aims at further delineating the steps and molecular triggers that allow the mature mRNA to reach the nucleopore and its subsequent translocation to the cytoplasm in normal and pathological cellular backgrounds.
Recent studies
- Mechanisms leading to RNA export factors recruitment and how this shapes the human transcriptome.
References used:
Preceding references: 1-5
6. Green, D. M. et al. Nab2p is required for poly(A) RNA export in Saccharomyces cerevisiae and is regulated by arginine methylation via Hmt1p. J. Biol. Chem. 277, 7752–7760 (2002).
7. Hurt, J. A. et al. A conserved CCCH-type zinc finger protein regulates mRNA nuclear adenylation and export. The Journal of Cell Biology 185, 265–277 (2009).
8. Strässer, K. & Hurt, E. Yra1p, a conserved nuclear RNA-binding protein, interacts directly with Mex67p and is required for mRNA export. EMBO J 19, 410–420 (2000).
9. Huang, Y., Gattoni, R., Stévenin, J. & Steitz, J. A. SR Splicing Factors Serve as Adapter Proteins for TAP-Dependent mRNA Export. Molecular Cell 11, 837–843 (2003).
10. Zolotukhin, A. S. U2AF Participates in the Binding of TAP (NXF1) to mRNA. J. Biol. Chem. 277, 3935–3942 (2001).
11. Hautbergue, G. M., Hung, M.-L., Golovanov, A. P., Lian, L.-Y. & Wilson, S. A. Mutually exclusive interactions drive handover of mRNA from export adaptors to TAP. Proc. Natl. Acad. Sci. U.S.A. 105, 5154–5159 (2008).
12. Ruepp, M., Aringhieri, C. & Vivarelli, S. Mammalian pre-mRNA 3' End Processing Factor CF Im68 Functions in mRNA Export. Molecular Biology of the Cell (2009).
13. Hautbergue, G. M. et al. UIF, a New mRNA export adaptor that works together with REF/ALY, requires FACT for recruitment to mRNA. Curr. Biol. 19, 1918–1924 (2009).
14. Saguez, C., Olesen, J. R. & Jensen, T. H. Formation of export-competent mRNP: escaping nuclear destruction. Current Opinion in Cell Biology 17, 287–293 (2005).
15. Schmid, M. & Jensen, T. H. Quality control of mRNP in the nucleus. Chromosoma 117, 419–429 (2008).
16. Stutz, F. et al. REF, an evolutionary conserved family of hnRNP-like proteins, interacts with TAP/Mex67p and participates in mRNA nuclear export. RNA 6, 638–650 (2000).